Peptide reconstitution explained for researchers: solvents, step-by-step methods, concentration formulas, storage stability, and common errors to avoid in 2026.
Updated at:Peptide reconstitution is the process of dissolving lyophilized (freeze-dried) peptide powder into a compatible solvent to create a stable liquid solution for research use. Proper reconstitution technique directly affects peptide integrity, concentration accuracy, and the reproducibility of experimental results. This guide covers the science behind lyophilization, solvent selection based on peptide properties, step-by-step reconstitution methods, concentration calculations, and post-reconstitution storage, all grounded in published research and laboratory best practices.
What Is Peptide Reconstitution and Why Does It Matter in Research?
Peptide reconstitution refers to the controlled addition of a solvent to a lyophilized peptide to restore it to a usable liquid form. Research-grade peptides are almost universally supplied as lyophilized powders because the freeze-dried state dramatically extends shelf life and preserves bioactivity during shipping and storage.
The reconstitution step is where many experimental variables are introduced. Incorrect solvent choice, excessive agitation, or imprecise volume measurements can degrade the peptide, alter its concentration, or introduce contaminants that compromise downstream assays. According to recommendations published in Clinical Proteomics, standardized peptide handling, including reconstitution, is essential for reproducible results in mass spectrometry-based assays and other quantitative methods. These guidelines have not been universally adopted across all research settings, highlighting the need for clear, accessible reconstitution resources.
For researchers working with peptides like BPC-157, TB-500, or GH secretagogues, the reconstitution step is not merely preparatory; it is a critical quality control point that determines whether the peptide retains its structural and functional properties throughout the study period.
Why Lyophilization Matters for Peptide Research
Lyophilization, commonly known as freeze-drying, is the standard method for stabilizing peptides for long-term storage. The process removes water from a frozen peptide solution through sublimation (ice converting directly to vapor under vacuum), leaving behind a dry, porous cake or powder.
The primary advantage of lyophilization is that it eliminates the aqueous environment where most degradation reactions occur. Research published in Interface Focus identified several factors that affect the physical stability of peptide therapeutics, including hydrolysis, deamidation, and oxidation, all of which are accelerated in aqueous solutions. By removing water, lyophilization slows these pathways substantially. These stability findings come from controlled laboratory studies and may vary depending on the specific peptide sequence and formulation conditions.
Three primary degradation pathways are relevant to reconstitution decisions:
Degradation Pathway | Susceptible Residues | Trigger | Relevance to Reconstitution |
|---|---|---|---|
Hydrolysis | Aspartate (Asp), Asparagine (Asn) | Water, elevated pH | Solvent pH and volume directly affect hydrolysis rate |
Oxidation | Cysteine (Cys), Methionine (Met), Tryptophan (Trp) | Oxygen, light, DMSO | Solvent choice and storage conditions matter |
Deamidation | Glutamine (Gln) at N-terminus | Time, pH, temperature | Post-reconstitution stability window is limited |
Lyophilized peptides stored at -20°C to -80°C can remain stable for 12 to 24 months or longer, depending on the sequence and storage conditions. Once reconstituted, however, stability drops significantly, with most peptide solutions maintaining integrity for 28 to 90 days under refrigeration when prepared with bacteriostatic water. This stability data is based on specific peptide formulations studied under controlled conditions and may not apply uniformly to all peptides.
Choosing the Right Solvent for Peptide Reconstitution
Solvent selection is not one-size-fits-all. The amino acid composition, net charge, and hydrophobicity of a peptide determine which solvent will achieve complete dissolution without damaging the molecule.
Bacteriostatic Water (Standard Solvent)
Bacteriostatic water containing 0.9% benzyl alcohol is the most commonly used solvent for peptide reconstitution in research settings. The benzyl alcohol serves as a preservative, inhibiting microbial growth and allowing multiple withdrawals from a single vial over a period of up to 28 days.
Most hydrophilic peptides, including those with a high proportion of charged amino acids (Lys, Arg, Glu, Asp), dissolve readily in bacteriostatic water. This covers the majority of research peptides in common use.
Sterile Water and Saline Solutions
Sterile water for injection contains no preservative and must be used immediately or within a single session. Phosphate-buffered saline (PBS) is used when maintaining physiological pH and ionic strength is important for the experiment. According to Pacific Immunology's reconstitution guidelines, most peptides are hydrophilic and will dissolve in saline solutions, PBS, or water without difficulty.
Acetic Acid Solutions
Peptides with a net positive charge (basic peptides rich in Lys, Arg, or His) that resist dissolution in neutral water can often be solubilized using dilute acetic acid (0.1% to 10%). The acid protonates basic residues, increasing solubility. Protide Health offers acetic acid solution specifically for this application in research contexts.
DMSO and Organic Solvents
Hydrophobic peptides with a high proportion of nonpolar residues (Ala, Val, Leu, Ile, Phe, Trp) may require dimethyl sulfoxide (DMSO), dimethylformamide (DMF), or acetonitrile (ACN) for initial dissolution. A 2024 protocol published in STAR Protocols describes a standardized method for reconstituting peptides from DMSO to aqueous buffers, confirming that DMSO can solubilize peptides at concentrations of 20 to 50 mM. However, DMSO is not compatible with peptides containing cysteine (Cys) or methionine (Met) residues, as it can promote oxidation of these amino acids.
Solvent Selection Decision Framework
Peptide Characteristic | Recommended First Solvent | Second Option |
|---|---|---|
Hydrophilic, neutral or mixed charge | Bacteriostatic water | Sterile water or PBS |
Basic (net positive charge, rich in Lys/Arg) | 0.1% acetic acid | Bacteriostatic water at lower pH |
Acidic (net negative charge, rich in Glu/Asp) | Dilute ammonium hydroxide (NH4OH) | PBS at pH 7.4 |
Hydrophobic (high Ala/Val/Leu/Ile/Phe content) | DMSO (small volume first) | DMF or ACN |
Contains Cys or Met residues | Bacteriostatic water (not DMSO) | Degassed PBS |
"Peptide sequences containing Cys and Met are unstable in DMSO due to oxidation. For these peptides, aqueous solvents or alternative organic solvents such as DMF should be used." Source: Bachem Technical Notes on Peptide Solubility
This solvent selection data is derived from manufacturer guidelines and published research protocols. Researchers should verify compatibility with their specific peptide and experimental conditions.
Step-by-Step Peptide Reconstitution for Research Applications
The following reconstitution method reflects standard laboratory practices documented across multiple published research protocols and manufacturer guidelines.

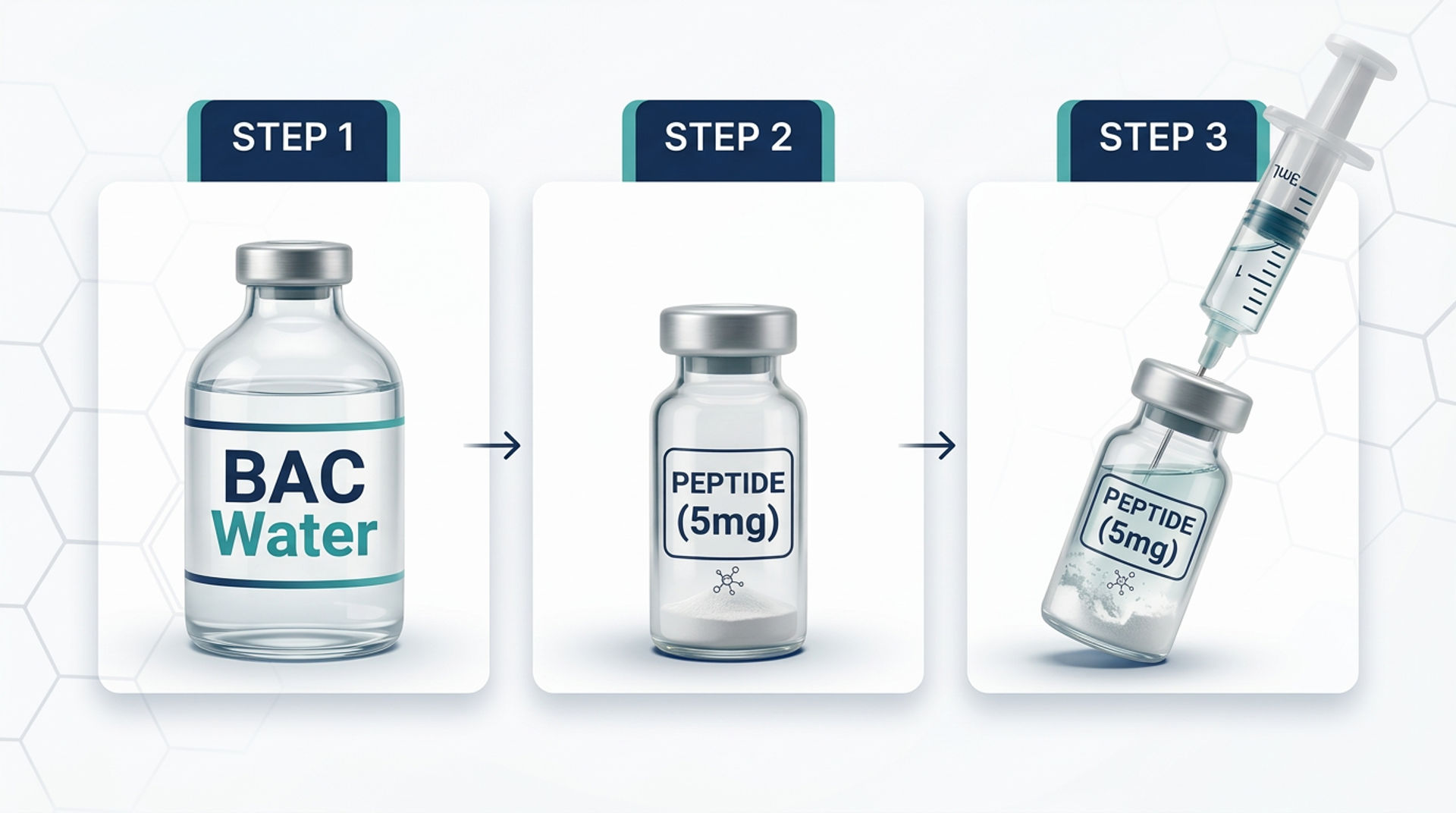
Using a fresh 3 mL syringe, remove the cap from the BAC water and draw out 2–3 mL. Then remove the cap from the peptide vial and insert the needle through the rubber stopper, letting the liquid flow gently down the side of the vial whenever possible. Once added, gently stir or swirl to reconstitute. Discard the syringe after use, and never reuse it to reconstitute other peptide vials.
Materials Required
Before beginning, gather: the lyophilized peptide vial, the chosen solvent (typically bacteriostatic water), sterile syringes (1 mL or 3 mL), sterile needles (18-21 gauge for drawing solvent, 25-30 gauge for transfer), alcohol prep pads, and a clean, flat workspace.
Step 1: Equilibrate to Room Temperature
Remove the lyophilized peptide vial from cold storage and allow it to reach room temperature (approximately 15 to 20 minutes). Opening a cold vial in a warmer environment can cause condensation to form inside the vial, introducing unwanted moisture to the peptide powder. Research recommendations suggest equilibrating in a desiccator to prevent water absorption during this step.
Step 2: Sterilize the Vial Stoppers
Using an alcohol prep pad, thoroughly wipe the rubber stopper on both the peptide vial and the solvent vial. Allow the alcohol to evaporate fully (approximately 30 seconds) before proceeding. This step prevents microbial contamination of the solution.
Step 3: Draw the Calculated Solvent Volume
Using a sterile syringe and needle, draw the predetermined volume of solvent. The volume depends on the desired final concentration (see the Calculations section below). For most research applications, 1 to 2 mL of bacteriostatic water per vial is standard.
Step 4: Add Solvent Slowly Along the Vial Wall
Insert the needle through the rubber stopper of the peptide vial at an angle. Aim the needle tip at the glass wall of the vial, not directly at the lyophilized powder. Depress the plunger slowly, allowing the solvent to run down the inside wall of the vial.
This technique matters: directing solvent onto the powder can cause foaming, which introduces air bubbles and can physically damage the peptide through shear forces at the air-liquid interface. Published research in Interface Focus confirms that agitation-induced aggregation is a documented degradation pathway for peptides in solution. These aggregation findings were observed under specific experimental conditions and may vary by peptide type.
Step 5: Dissolve by Gentle Swirling
Once all solvent has been added, gently swirl the vial by rotating it between your palms. Alternatively, tilt the vial at a 45-degree angle and slowly roll it. The powder should dissolve within 1 to 5 minutes for most peptides. According to JPT Peptide Technologies, allowing 15 to 30 minutes for incubation can facilitate complete dissolution of slower-dissolving sequences.
Never shake the vial vigorously. Shaking creates foam, introduces air (which promotes oxidation), and can cause mechanical degradation of the peptide chain.
Step 6: Inspect the Solution
The reconstituted solution should be completely clear and free of visible particles, cloudiness, or foam. A cloudy solution indicates incomplete dissolution or aggregation. If cloudiness persists after gentle swirling, the peptide may require a different solvent (refer to the solvent selection framework above) or a brief period of gentle sonication.

"If the solution has gelled, is cloudy, or contains visible particles, this indicates that the peptide has not been completely dissolved and may require a different solvent system or additional dissolution time." Source: The Chemical Record, 2024
Reconstitution Calculations and Concentration Formulas
Accurate concentration calculations ensure that each withdrawal from the reconstituted vial delivers a consistent, known amount of peptide. The fundamental reconstitution formula is straightforward:
Concentration (mg/mL) = Amount of Peptide (mg) / Volume of Solvent (mL)
Worked Example
For a vial containing 5 mg of peptide reconstituted with 2 mL of bacteriostatic water:
5 mg / 2 mL = 2.5 mg/mL (or 2,500 mcg/mL)
If a research application calls for 250 mcg per use, you would draw:
250 mcg / 2,500 mcg/mL = 0.1 mL (or 10 units on a standard 100-unit insulin syringe)
Common Reconstitution Concentrations
Vial Content | Solvent Volume | Resulting Concentration | 250 mcg Draw Volume |
|---|---|---|---|
5 mg | 1 mL | 5 mg/mL | 0.05 mL (5 units) |
5 mg | 2 mL | 2.5 mg/mL | 0.1 mL (10 units) |
10 mg | 1 mL | 10 mg/mL | 0.025 mL (2.5 units) |
10 mg | 2 mL | 5 mg/mL | 0.05 mL (5 units) |
10 mg | 3 mL | 3.33 mg/mL | 0.075 mL (7.5 units) |
Adding more solvent does not change the total amount of peptide in the vial; it only changes the concentration per unit volume. This is a common point of confusion: the peptide quantity is fixed by the vial content, and solvent volume determines how concentrated or dilute the solution is.
Peptide Mind's peptide dosage calculator automates these calculations, including unit conversions between mg, mcg, mL, and syringe units.
Storage and Stability After Reconstitution
Once reconstituted, peptides enter a less stable state than their lyophilized form. Proper storage is essential to maintain peptide integrity throughout the research period.
Refrigeration (2°C to 8°C)
Store reconstituted peptide solutions in the refrigerator immediately after preparation. Most peptides reconstituted with bacteriostatic water remain stable for 28 to 90 days under continuous refrigeration. A study on lyophilized teriparatide (PTH 1-34) published in the Journal of Pharmaceutical Sciences demonstrated that reconstituted peptide solutions maintained stability over a 28-day refrigerated period at the studied concentrations. Stability timelines vary by peptide, and researchers should consult peptide-specific data when available.
Avoid Repeated Freeze-Thaw Cycles
If a reconstituted solution must be frozen, divide it into single-use aliquots before freezing. Each freeze-thaw cycle exposes the peptide to ice crystal formation, which can cause physical damage, aggregation, and loss of bioactivity. Research published in Pharmaceutics identifies freeze-thaw cycling as a significant contributor to peptide degradation in solution. These observations were made under controlled conditions using specific peptide formulations.
Protect from Light
Many peptides, particularly those containing tryptophan (Trp) or tyrosine (Tyr) residues, are susceptible to photodegradation. Store reconstituted vials in a dark location or wrap them in aluminum foil. Sigma-Aldrich's peptide stability guidelines confirm that light exposure accelerates oxidative degradation pathways in susceptible sequences.
Key Storage Parameters
Parameter | Lyophilized (Unreconstituted) | Reconstituted (BAC Water) |
|---|---|---|
Recommended Temperature | -20°C to -80°C | 2°C to 8°C (refrigerator) |
Typical Stability Period | 12 to 24+ months | 28 to 90 days |
Light Sensitivity | Low (solid state) | Moderate to High (in solution) |
Humidity Sensitivity | High (keep desiccated) | N/A (already in solution) |
Multi-Access | N/A | Yes, with BAC water (up to 28 days per USP guidelines) |
"The rate of chemical degradation approximately doubles for every 10°C increase in storage temperature for most peptide solutions, making strict temperature control during storage a primary factor in maintaining research material integrity." Source: Sigma-Aldrich Peptide Stability Technical Document
Two-Step Reconstitution for Difficult Peptides
Some peptides resist dissolution in any single aqueous solvent due to their amino acid composition. These are typically sequences with a high proportion of hydrophobic residues (Ala, Val, Leu, Ile, Phe, Trp) or peptides with mixed hydrophobic and charged regions that create amphipathic structures. For these peptides, a two-step reconstitution approach using an organic co-solvent followed by aqueous dilution is the standard method documented in published research protocols.
When to Use Two-Step Reconstitution
Two-step reconstitution is indicated when the peptide does not dissolve within 30 minutes of gentle swirling in aqueous solvent, when the solution remains visibly cloudy or contains particulate matter after initial reconstitution, or when the peptide's certificate of analysis or manufacturer documentation specifically recommends organic co-solvent use. Research published in The Chemical Record documents the challenges of solubilizing amyloid-forming and hydrophobic peptide sequences, confirming that organic co-solvents are often necessary for complete dissolution.
Two-Step Method
First, add a small volume (typically 50 to 100 microliters) of DMSO or DMF directly to the lyophilized peptide. Swirl gently until the powder is fully dissolved in the organic solvent. This creates a concentrated stock solution. Second, slowly add the aqueous solvent (bacteriostatic water or PBS) to the desired final volume while gently swirling. The aqueous addition should be gradual to prevent the peptide from crashing out of solution as the solvent polarity changes. A protocol published in STAR Protocols provides a standardized method for this DMSO-to-aqueous transition, including recommended dilution ratios and mixing techniques.
Important Considerations for Two-Step Reconstitution
When using DMSO as the initial solvent, researchers should note that DMSO is cytotoxic at concentrations above 1% in cell-based assays. Planning the dilution step to keep final DMSO concentration below this threshold is essential for maintaining assay validity. Additionally, DMSO has a relatively high freezing point (18.5°C), meaning solutions stored in the refrigerator may partially solidify, which can complicate subsequent withdrawals. Bringing the vial to room temperature before each use addresses this issue.
The two-step method adds a step to the workflow but significantly expands the range of peptides that can be successfully reconstituted for research use. Documentation of the exact solvent volumes, concentrations, and dilution ratios used is critical for reproducibility across experiments.
Common Reconstitution Errors and How to Avoid Them
Reconstitution errors can compromise peptide integrity and lead to unreliable research data. These are the most frequently documented issues in laboratory settings:
Shaking the vial instead of swirling. Vigorous shaking creates foam and air-liquid interfaces that promote peptide aggregation. Aggregated peptides lose bioactivity and can produce inconsistent results. Always swirl gently or roll the vial between your palms.
Injecting solvent directly onto the powder. Directing the solvent stream onto the lyophilized cake can cause localized high concentrations and foaming. Aim the needle at the glass wall and allow solvent to run down slowly.
Using the wrong solvent. Attempting to dissolve a hydrophobic peptide in water alone will result in an incomplete, cloudy solution. Similarly, using DMSO for peptides containing cysteine or methionine can cause oxidation. Refer to the solvent selection framework and the peptide's certificate of analysis for guidance.
Opening a cold vial in a warm environment. Condensation introduces uncontrolled water into the vial, which can partially dissolve the peptide unevenly and alter the final concentration. Always equilibrate to room temperature first.
Inaccurate volume measurement. Small measurement errors have an outsized impact when working with milligram quantities. Use calibrated syringes and draw solvent at eye level to ensure accuracy. Peptide Mind's dosage calculator can verify your calculations before reconstitution.
Storing reconstituted peptides at room temperature. Reconstituted solutions left at ambient temperature degrade at approximately twice the rate compared to refrigerated storage for each 10°C increase. Refrigerate immediately after preparation.
Frequently Asked Questions
What volume of bacteriostatic water is typically used in peptide reconstitution research?
The most common reconstitution volumes in published research protocols range from 1 mL to 3 mL of bacteriostatic water per vial, depending on the peptide quantity and the desired working concentration. For a 5 mg peptide vial, 2 mL of bacteriostatic water produces a 2.5 mg/mL concentration, which is practical for most research applications using standard syringes. The specific volume does not change the total peptide amount; it only affects the concentration per draw. Further research into optimal reconstitution volumes for specific peptide families remains an active area of investigation.
How long does peptide reconstitution take in a laboratory setting?
Most hydrophilic peptides dissolve within 1 to 5 minutes of gentle swirling after solvent addition. Some peptides, particularly those with hydrophobic regions or larger molecular weights, may require 15 to 30 minutes of gentle incubation at room temperature for complete dissolution. If a peptide has not dissolved after 30 minutes of gentle swirling, this typically indicates that a different solvent or a two-step dissolution approach may be needed.
What is the standard reconstitution concentration formula?
The formula is: Concentration (mg/mL) = Peptide Amount (mg) / Solvent Volume (mL). For example, 10 mg of peptide reconstituted with 2 mL of bacteriostatic water yields a 5 mg/mL solution. To determine the draw volume for a specific amount, divide the desired amount by the concentration: 500 mcg / 5,000 mcg/mL = 0.1 mL, which equals 10 units on a 100-unit syringe.
Should lyophilized peptides be refrigerated before reconstitution?
Lyophilized peptides are best stored at -20°C to -80°C for long-term preservation. Before reconstitution, the vial should be brought to room temperature gradually (15 to 20 minutes) to prevent condensation from forming inside the vial. Opening a cold vial in a warm environment introduces moisture that can unevenly dissolve the peptide and affect final concentration accuracy. Research guidelines from Clinical Proteomics recommend equilibrating in a desiccator for optimal results.
How long are reconstituted peptides stable in research settings?
Reconstituted peptides prepared with bacteriostatic water and stored under continuous refrigeration (2°C to 8°C) generally maintain stability for 28 to 90 days, depending on the peptide sequence and concentration. Sterile water preparations without preservative should be used within a single session or within 24 hours. A stability study on lyophilized teriparatide confirmed 28-day stability under refrigerated conditions. However, stability varies considerably across different peptide sequences, and researchers should verify with peptide-specific data when available.
The Research Foundation for Proper Peptide Reconstitution
Peptide reconstitution is a foundational laboratory skill that directly influences the quality and reproducibility of peptide research. From solvent selection based on amino acid properties to precise concentration calculations and proper post-reconstitution storage, each step in the process has a measurable impact on peptide integrity.
The published literature consistently demonstrates that standardized reconstitution practices reduce variability and preserve bioactivity across research applications. For automated reconstitution calculations, Peptide Mind's peptide dosage calculator provides a free tool designed for researchers working with lyophilized peptides.
Researchers seeking lab-tested peptides and reconstitution supplies can explore Protide Health's peptide catalog for materials meeting research-grade standards.
References
Grant RP, Hoofnagle AN. "From lost in translation to paradise found: enabling protein biomarker method transfer by mass spectrometry." Clinical Chemistry, 60(7), 2014. PMC4830481
Roberts CJ. "Factors affecting the physical stability (aggregation) of peptide therapeutics." Interface Focus, 7(6), 2017. PMC5665799
Drucker DJ. "Strategies for Improving Peptide Stability and Delivery." Pharmaceutics, 14(11), 2022. PMC9610364
Grasso G, et al. "Protocol for reconstituting peptides/peptidomimetics from DMSO to aqueous buffers for circular dichroism analyses." STAR Protocols, 5(1), 2024. PMC10839526
Ohashi R, et al. "Stability of lyophilized teriparatide, PTH(1-34), after reconstitution." Journal of Pharmaceutical Sciences, 105(2), 2016. PubMed 26620825
Polańska E. "Challenges in Peptide Solubilization: Amyloids Case Study." The Chemical Record, 24(10), 2024. Wiley Online Library
Pacific Immunology. "Peptide Reconstitution." pacificimmunology.com
Sigma-Aldrich. "Peptide Stability and Potential Degradation Pathways." sigmaaldrich.com
Bachem. "Peptide Solubility Technical Notes." bachem.com
JPT Peptide Technologies. "How to Reconstitute Peptides." jpt.com
Research Disclaimer: The information presented in this article is for educational and research purposes only. Peptide Mind provides evidence-based research summaries and does not offer medical advice, diagnosis, or treatment recommendations. All peptides discussed are intended for in vitro and preclinical research use only. Consult a qualified healthcare professional before making any health-related decisions. The research cited may not reflect the full body of available evidence, and findings from preclinical studies may not translate to human outcomes.
Add a comment
This will be publicly visible.
Your email address will not be published.
Your comment will be reviewed by an admin before it is published.